pH Measurement: How it Makes a Difference
Measuring pH goes way beyond science class. For industries and researchers, ensuring accurate pH levels across products and resources is essential for protecting communities and supporting wellbeing.
Below, read about the basics of pH measurement.
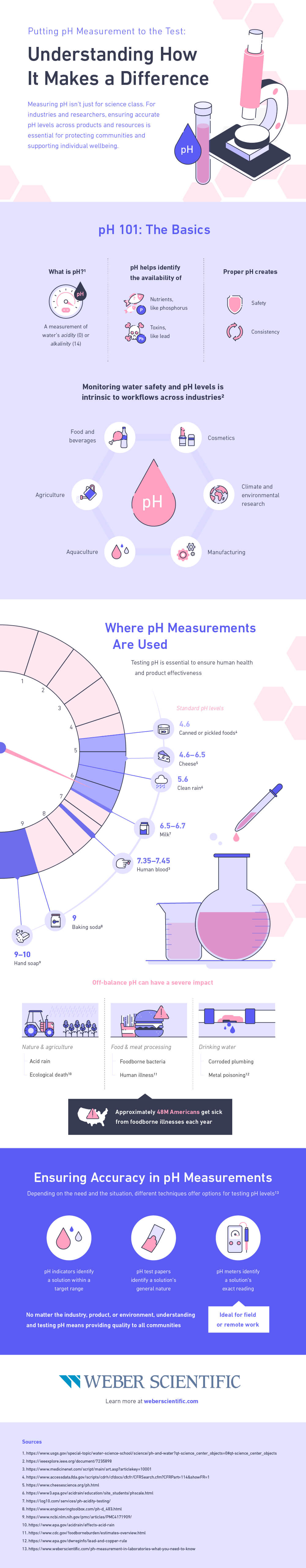
pH 101: The Basics
pH is a measurement of waters acidity or alkalinity. It helps identify the availability of nutrients like phosphorus and the presence of toxins like lead. Proper pH creates safety and consistency.
Monitoring water safety and pH levels is intrinsic to workflows across industries, including:
- Food and beverages
- Cosmetics
- Climate and environmental research
- Manufacturing
- Aquaculture
- Agriculture
Where pH Measurements Are Used
Approximately 48 million Americans get sick from foodborne illnesses each year. Testing pH is essential to ensure human health and product effectiveness.
Standard pH levels:
- 4.6: canned or pickled foods
- 4.6-6.5: cheese
- 5-6: clean rain
- 6.5-6.7: milk
- 7.35 - 7.45: human blood
- 9: baking soda
- 9-10: hand soap
Off-balance pH can have a severe impact:
- Nature and agriculture (acid rain, ecological death)
- Food and meat processing (food borne bacteria, human illness)
- Drinking water (corroded plumbing, metal poisoning)
Ensuring Accuracy in pH Measurements
Depending on the need and the situation, different techniques offer options for testing pH levels.
- pH indicators identify a solution within a target range
- pH test papers identify a solution’s general nature
- pH meters identify a solutions exact reading (ideal for field or remote work)
No matter the industry, product or environment, understanding and testing pH means providing quality to all communities.

